(hook tail)
Rev. 10/07/2021
Onchocerca volvulus
Onchocerca volvulus has, for many years, been regarded as the causative agent of River Blindness. Reports in 2002 indicate that the bacterium Wolbachia, which is associated with the nematodes and which may be required for their growth and reproduction, has a major role in the pathology of the disease. Wolbachia incites a severe inflammatory response, leading to blindness and serious skin disorders.
The immune response of the host is also involved in the disease. Mice lacking an immune cell receptor molecule called TLR4 showed fewer signs of eye inflammation when exposed to Wolbachia-laden worm extract (Hoerauf and Volkmann, 2002).
The programs of the Carter Center and the development of avermectin-based antheminthics has dramatically decreased the incidences of river blindness and lymphatic filariasis (elephantiasis).
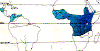
Human onchocerciasis is found in both the Old and New World but about 96% of all cases are in Africa and mostly in Western Africa. Of the 36 countries where the disease is endemic, 30 are in sub-Sahara Africa (plus Yemen) and six are in the Americas. Important foci exist also in Mexico, Guatemala, Venezuela and Ecuador. A total of 18 million people are infected with the disease and have dermal microfilariae, of whom 99% are in Africa. (WHO's Fact Sheets #95)
River blindness is the second leading infectious cause of blindness in the world. It is spread to humans by the bite of black flies infected with O. volvulus.
The female nematode lays eggs in the host blood stream and these enter fly when it bites the human, Changes into larval stage (the microfilariae) within 24 hours after fly has first blood meal. Spreads throughout hosts muscles (vector is the Simulium commonly known as "the black fly" - vector bites human and microfilariae enter through wound; gradually grow into adult stages; young can not grow until they enter vector first live mainly in skin tissues of humans (as adult forms) in hard coverings (nodules), but has also been found in urine, blood, eye tissue, and more rarely in hydrocele fluid and lymph nodes. Young inhabit vector known as "the black fly" (Simulium), live in stomach blood, and muscle tissues has developed long life span and can live up to 15 years, survives this long even though presence in human does cause an immune response to presence in host
Surgical removal of nematode from eye; chemotherapy.
Pinpointing bacteria as the direct factor behind the virulence suggests new therapies for combating river blindness, especially since recent studies in infected humans have shown that the bacteria can be killed by the common antibiotic doxycycline.
The battle against river blindness is taking place on two fronts at the moment:
Antibiotic treatment of Wolbachia may help reduce the severity of the symptoms of river blindness in already-infected individuals.
Targeting Wolbachia could prevent the spread of Onchocerca. Recent studies have shown that doxycycline treatments in infected humans kills Wolbachia and also sterilize the nematodes, breaking the life cycle. In contrast, ivermectin treatments only reduce numbers of microfilariae for a few months and repeated treatments are necessary.